The latter are said to reside in the second coordination sphere. Each coordinated water molecule may be attached by hydrogen bonds to other water molecules. The bond between a water molecule and the metal ion is a dative covalent bond, with the oxygen atom donating both electrons to the bond. 
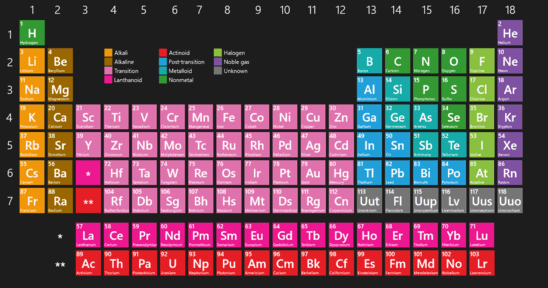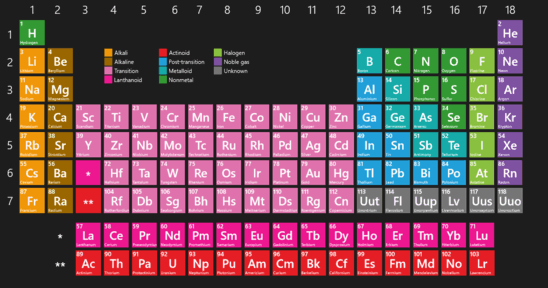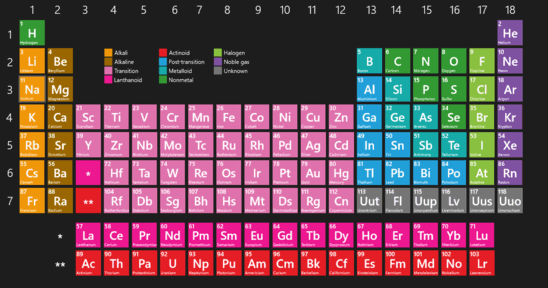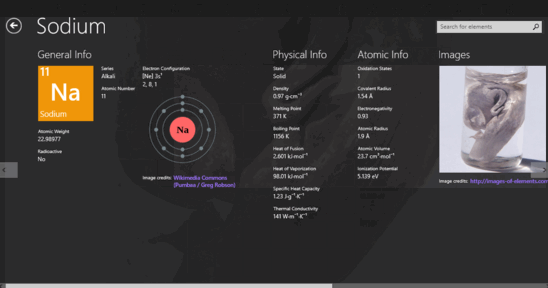
In aqueous solution the water molecules directly attached to the metal ion are said to belong to the first coordination sphere, also known as the first, or primary, solvation shell. Up to 12 water molecules may be present in the second shell (only two are shown in this diagram) linked by hydrogen bonds to the molecules in the first shell. First and second solvation shells of an octahedral aqua ion. The oxygen atoms are arranged at the vertices of a regular octahedron centered on the sodium ion. Schematic representation of the aqua ion +. For some highly radioactive elements, experimental chemistry has been done, and aqua cations may have been formed, but no experimental information is available regarding the structure of those putative aqua ions. The transactinides have been greyed out due to a lack of experimental data. Germanium is a semiconductor rather than a metal, but appears to form an aqua cation similarly, hydrogen forms an aqua cation like metals, despite being a gas. Some elements, such as tin and antimony, are clearly metals, but form only covalent compounds in the highest oxidation states: their aqua cations are restricted to their lower oxidation states. A few metallic elements that are commonly found only in high oxidation states, such as niobium and tantalum, are not known to form aqua cations near the metal–nonmetal boundary, arsenic and tellurium are only known as hydrolysed species. In the highest oxidation states only oxyanions, such as the permanganate(VII) ion, MnO −Ĥ, are known. With the higher oxidation states the simple aqua ions dissociate losing hydrogen ions to yield complexes that contain both water molecules and hydroxide or oxide ions, such as the vanadium(IV) species 2+. Compounds of the metallic elements usually form simple aqua ions with the formula z+ in low oxidation states. * No experimental information regarding aqua ion structures Introduction to metal aqua ions Elements that form aqua cations Aqua ions are prominent in electrochemistry. The residence time of a molecule in the first shell varies among the chemical elements from about 100 picoseconds to more than 200 years. Water molecules in the first hydration shell exchange with molecules in the second solvation shell and molecules in the bulk liquid. The aqua ion is associated, through hydrogen bonding with other water molecules in a secondary solvation shell. The logarithm of the first hydrolysis constant is proportional to z 2/ r for most aqua ions. The strength of the bonds between the metal ion and water molecules in the primary solvation shell increases with the electrical charge, z, on the metal ion and decreases as its ionic radius, r, increases. Lanthanide and actinide aqua ions have higher solvation numbers (often 8 to 9), with the highest known being 11 for Ac 3+. The solvation number, n, determined by a variety of experimental methods is 4 for Li + and Be 2+ and 6 for most elements in periods 3 and 4 of the periodic table. 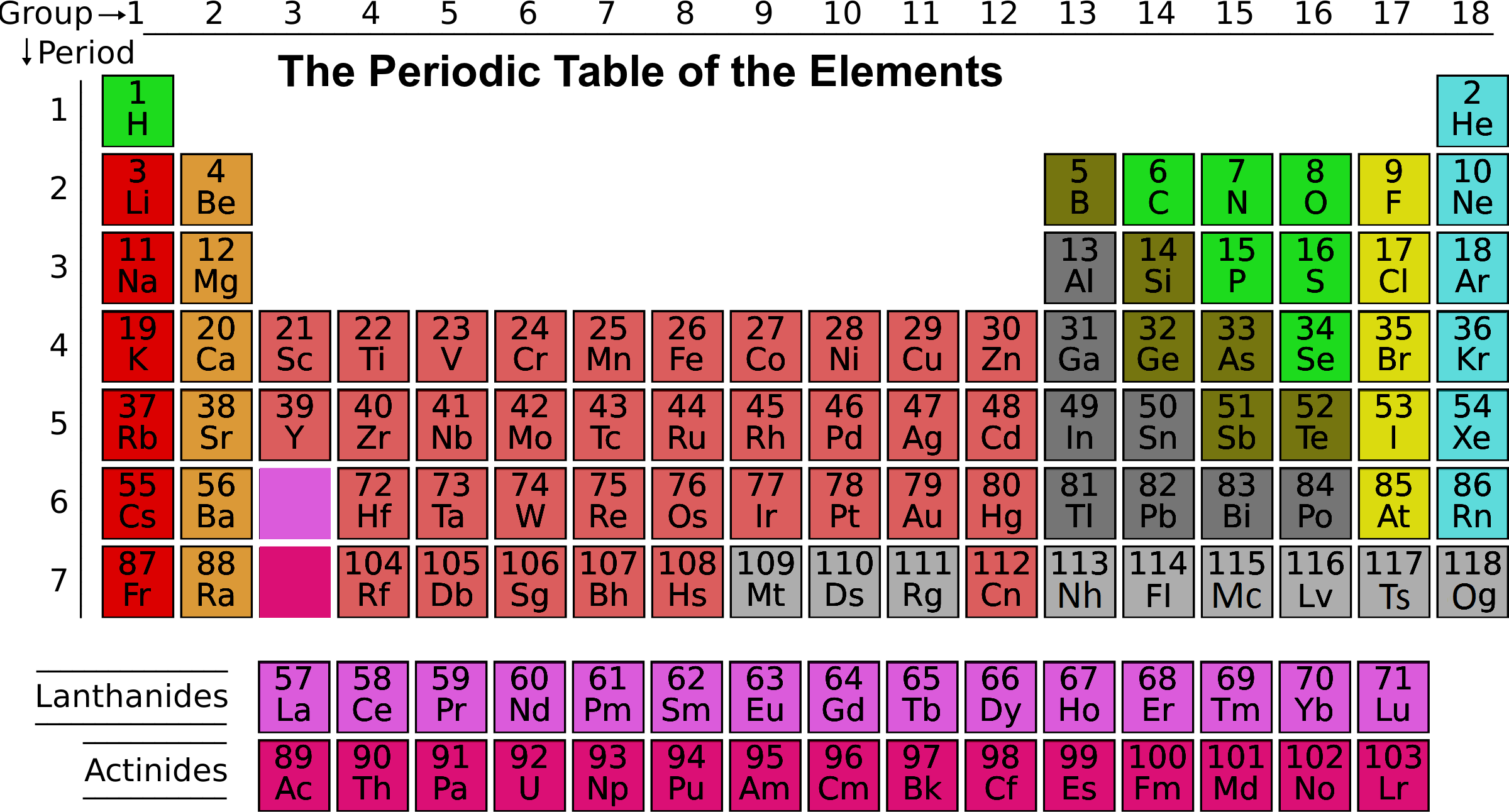
Properties and behavior of hydrated cations in aqueous solutionĪ metal ion in aqueous solution or aqua ion is a cation, dissolved in water, of chemical formula z+.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
