|
Revert™ 700 Total Protein Stain Kit and Revert 520 Total Protein Stain Kit ( /revertkit) Protein concentration must be determined for all samples. This protocol is intended for relative comparison of pan-protein and phospho-protein signals, and results do not indicate the stoichiometry of phosphorylation (1). 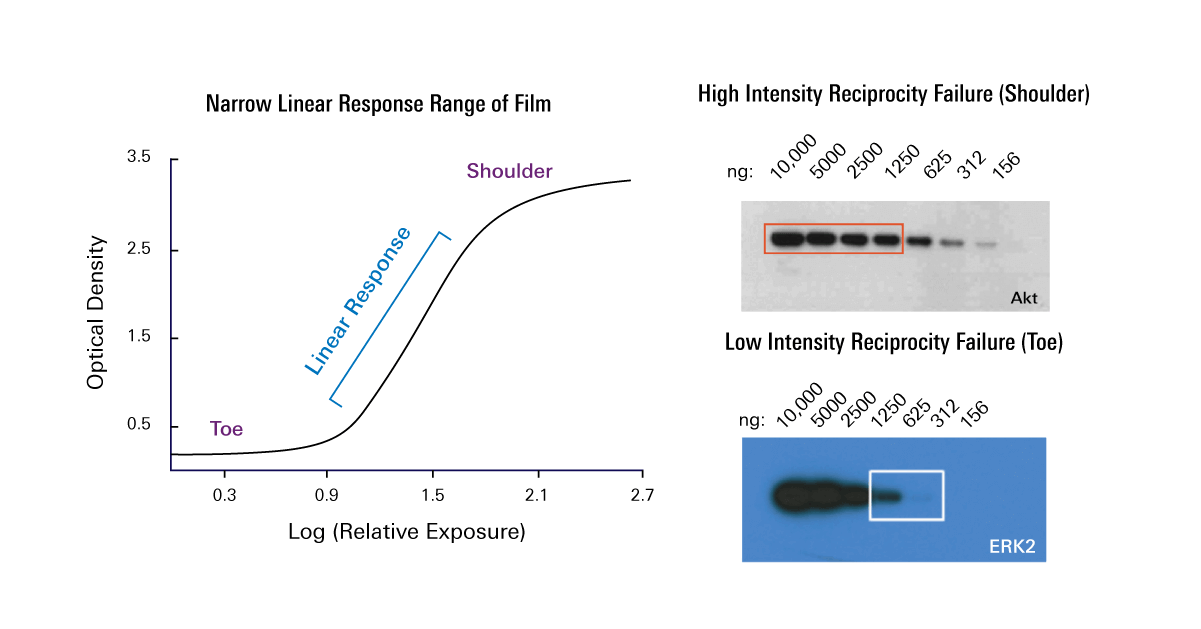
Important guidelines are provided in Section Verify specificity of the phospho-antibody to ensure that it does not cross-react with the unmodified target protein, and to identify possible interference from background bands. The Antibody Publication Database can help you find antibody pairs that work for your experiment ( /antibodyrequest).Īntibody validation. Always perform single-color control blots first to verify antibody specificity, and to identify possible interference from background bands. 
Two-color Western blot detection requires careful selection of primary and secondary antibodies to prevent cross-reactivity. However, these reagents do not preclude the need to perform a protein concentration assay before sample preparation and loading.Īntibody validation. You can use reagents designed to confirm uniform sample loading, such as Odyssey Loading Indicators (P/N 926-20002), to improve the accuracy of this validation protocol. A protein concentration assay (BCA, Bradford, or similar assay) must be used to adjust sample concentration and load all samples as consistently as possible. Uniform loading of total sample protein across the gel is critical for accurate QWB analysis. Replicates are discussed further on page Normalization Calculations and Analysis of Replicates. A minimum of three technical replicates is recommended for each sample. Replicate samples provide information about the inherent variability of your methods, to determine if the changes you see are meaningful and significant. See the protocol: Determining the Linear Range for Quantitative Western Blot Detection ( /LinearRange) for more information. Use a dilution series to verify that you are working within the linear range of detection, and signal intensity is proportional to sample loading. Saturated bands and sample overloading frequently compromise the accuracy of QWB. The Odyssey M Imager is the only Odyssey Imager that supports the 520 channel. This protocol describes how to use Revert 700 Total Protein Stain and Revert 520 Total Protein Stain for Western blot normalization and quantitative analysis. Revert staining is imaged at 700 nm or 520 nm, and fluorescent signals are proportional to sample loading. Revert™ 700 Total Protein Stain and Revert 520 Total Protein Stain ( /revert) are near-infrared fluorescent membrane stains used for total protein detection and normalization. This antibody-independent method corrects for variation in both sample protein loading and transfer efficiency, and monitors protein transfer across the blot at all molecular weights. 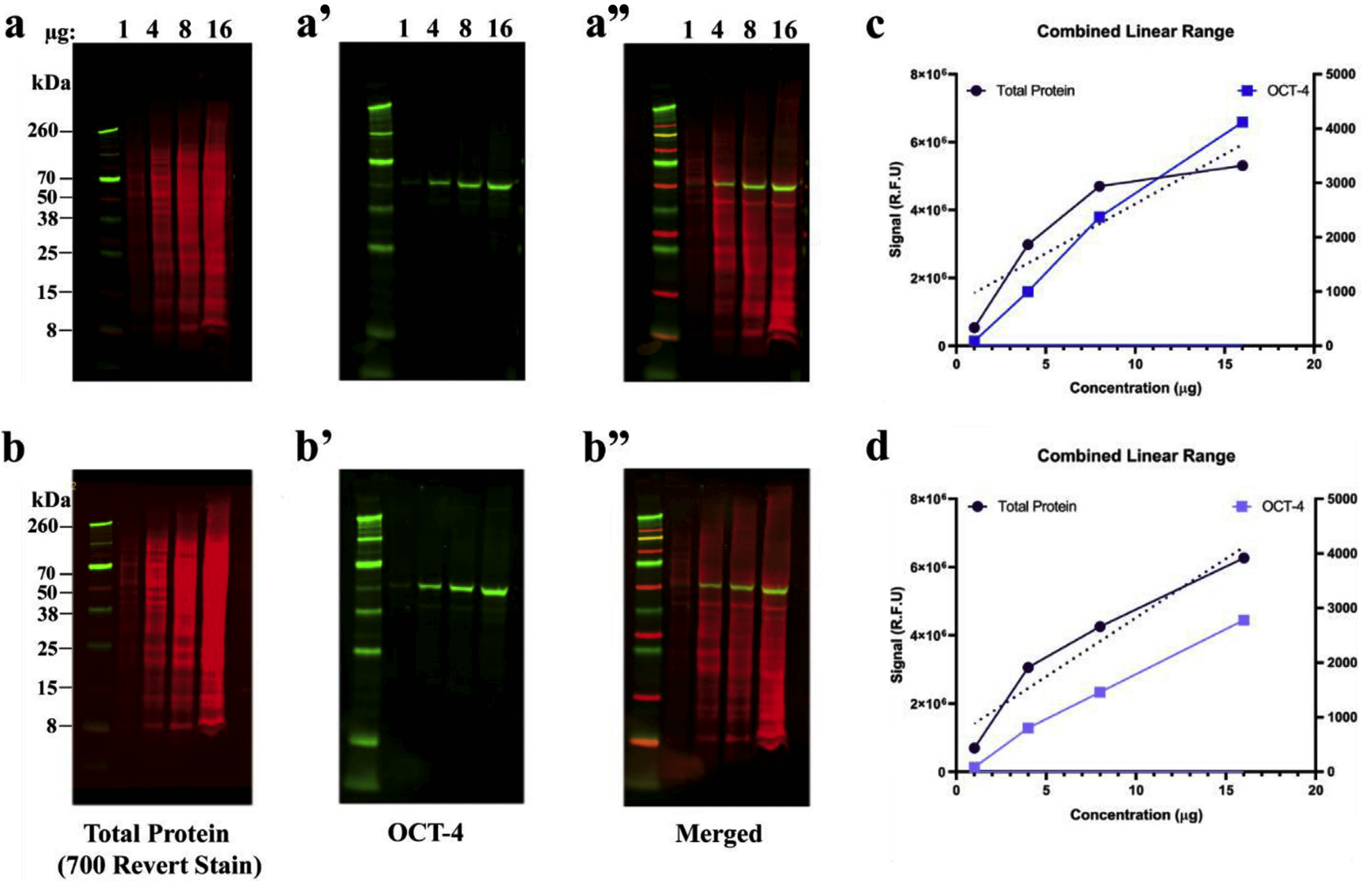
Because this internal loading control uses the combined signal from many different sample proteins in each lane, error and variability are minimized. After transfer, but prior to immunodetection, the membrane is treated with a total protein stain to assess actual sample loading across the blot. Total protein detection is becoming the “gold standard” for normalization of protein loading. Total Protein Staining as an Internal Loading Control The internal loading control is used as an indicator of sample protein loading, to correct for loading variation and confirm that observed changes represent actual differences between samples.įor more normalization related resources, see " Further Reading". In quantitative Western blotting (QWB), normalization mathematically corrects for unavoidable sample-to-sample and lane-to-lane variation by comparing the target protein to an internal loading control. Revert™ Total Protein Stain Normalization Protocol Introduction
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
